Review article in J Mol Cell Biol: Non-Homologous functions of the AlkB Homologs an update of the present knowledge

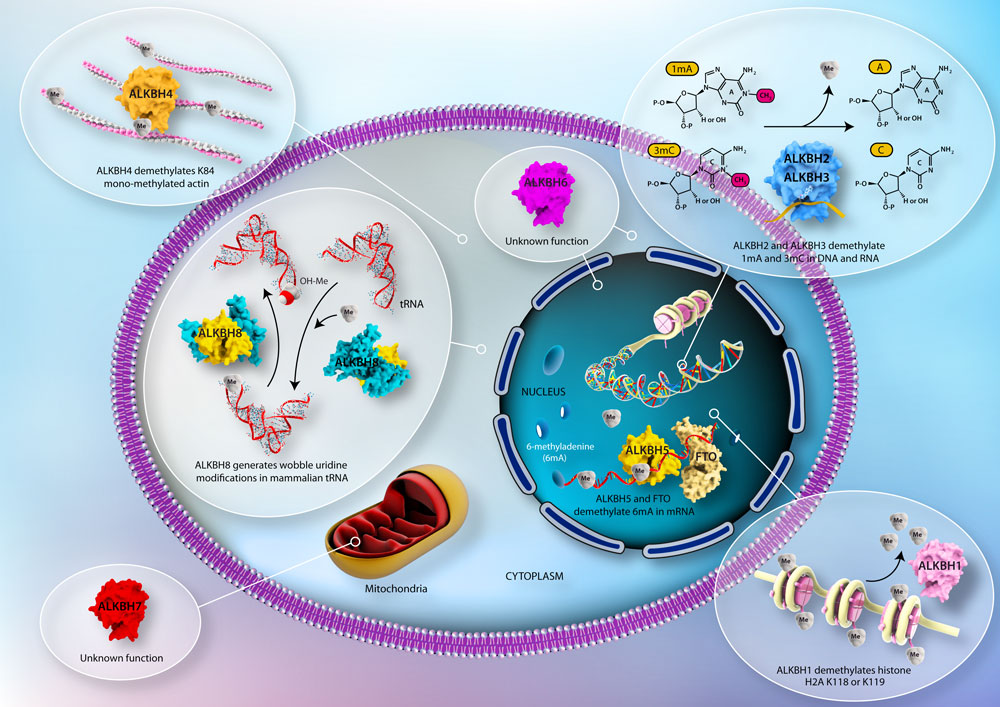
The DNA repair enzyme AlkB was identified in E. coli more than three decades ago. In E. coli the alkB gene encodes an enzyme primarily reversing the lesions 1-methyladenine and 3-methylcytosine in DNA. Since then, nine mammalian homologs, all members of the superfamily of alpha-ketoglutarate and Fe(II)-dependent dioxygenases, have been identified (designated ALKBH1-8 and FTO).
While E. coli AlkB serves as a DNA repair enzyme, only two mammalian homologs have been confirmed to repair DNA in vivo. The other mammalian homologs have remarkably diverse substrate specificities and biological functions. Substrates recognized by the different AlkB homologs comprise erroneous methyl- and etheno adducts in DNA, unique wobble uridine modifications in certain tRNAs, methylated adenines in mRNA and methylated lysines on proteins. The phenotypes of organisms lacking or overexpressing individual AlkB homologs include obesity, severe sensitivity to inflammation, infertility, growth retardation and multiple malformations.
The mammalian AlkB homologs have been extensively studied by scientists at the Department of Microbiology, particularly by the groups led by Professor Arne Klungland and Dr. Elisabeth Larsen. Primary in vivo substrates for ALKBH2 and 3 was published in 2006 (Ringvoll et al. EMBO J. 2006), and the requirement of ALKBH8 for certain tRNA wobble uridine modifications was shown in 2010 (Songe-Møller et al. Mol. Cell Biol. 2010). Internal mRNA methylations is reversible by ALKBH5 and FTO (Zheng and Dahl et al. Mol. Cell 2013; Jia et al. Nature Chem. Biol. 2011) and ALKBH1 plays a role during embryonic stem cell differentiation and epigenetic regulation through activity on histone H2A (Ougland et al. Stem Cells 2012). ALKBH4 is required for proper acin-myosin interaction (Li and Nilsen et al. Nature Comm. 2013) while lack of ALKBH7 leads to obesity (Solberg et al. Journal of Mol. Cell Biol. 2013). The only AlkB homolog left mysterious is the ALKBH6 protein. See the figure overview from the paper.
Taken together; the family of mammalian AlkB homologs is required for a broad range of biochemical reactions in a mammalian cell. A recent review in the prestigious Journal of Molecular Cell Biology (impact factor 8.43) by Ougland, Rognes, Klungland and Larsen at the Department of Microbiology describes the present knowledge of the mammalian AlkB homologs and their implications for human disease and development.
 |
| (click to enlarge) |
Links:
Non-homologous functions of the AlkB homologs.
Ougland R, Rognes T, Klungland A, Larsen E.
J Mol Cell Biol. 2015 May 23. pii: mjv029. [Epub ahead of print] Review.
Elisabeth Larsens group group: Laboratory of Embryonic Stem Cell Research
Arne Klunglands group: Laboratory for Genome repair and regulation
