Ongoing research projects
The research group works on developing new cell-based therapies to treat diabetes in a pre-clinical and clinical setting.
- Clinical trials
- Clinical beta cell replacement program (allogenic/autologous transplantation of beta cells)
- Generate new functional beta cells from pluripotent stem cells and understand their metabolism
- Developing Advanced Therapy Medicinal Products (ATMPs). These are a class of medical products that include gene therapies, cell therapies, and tissue-engineered products.
- Development of a novel cell therapy based on decidual stromal cells for the treatment of type 1 diabetes
- Developing organ-on-chip platform with pancreas and liver for disease modelling and drug screening
More information to come..
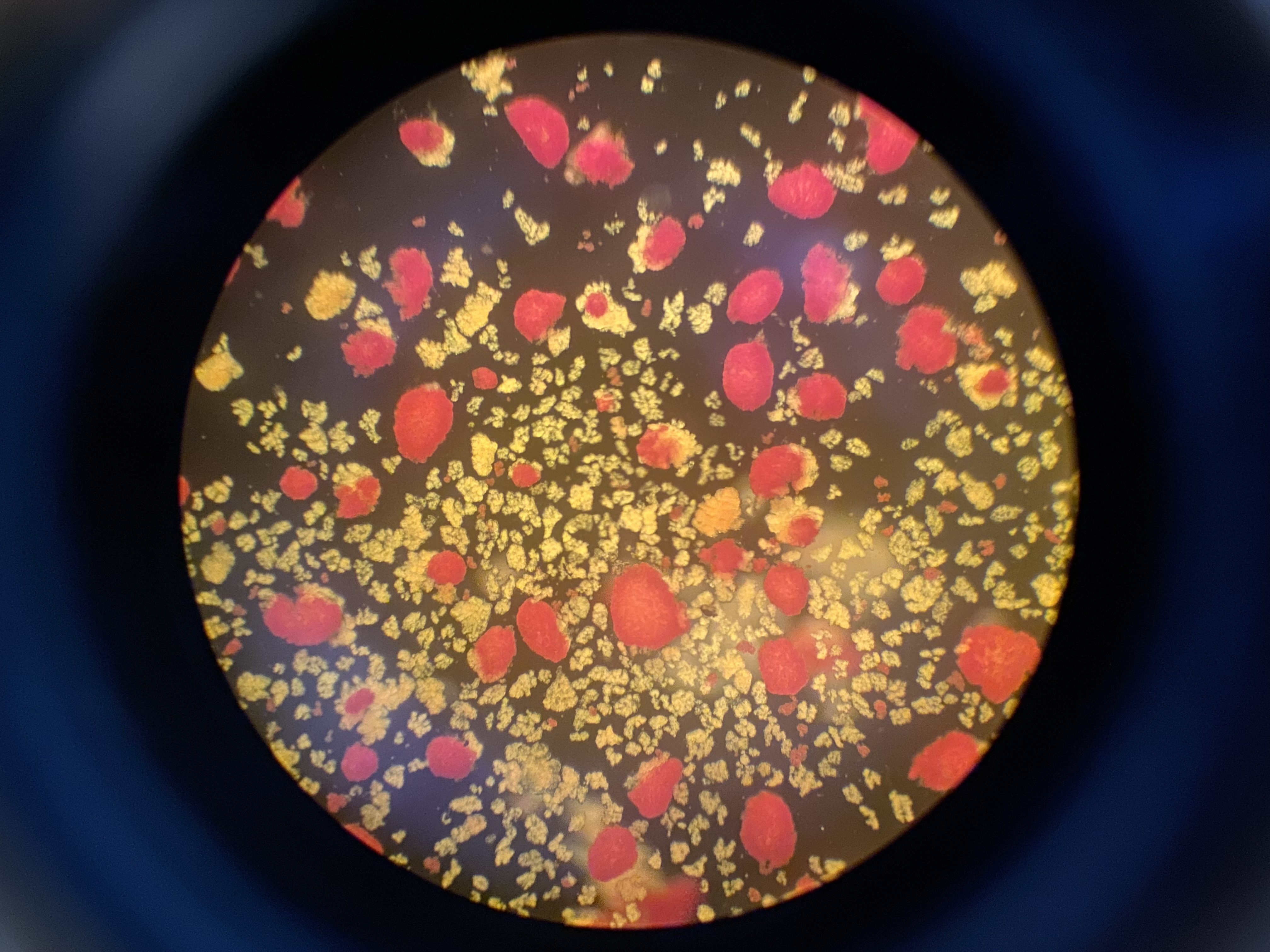
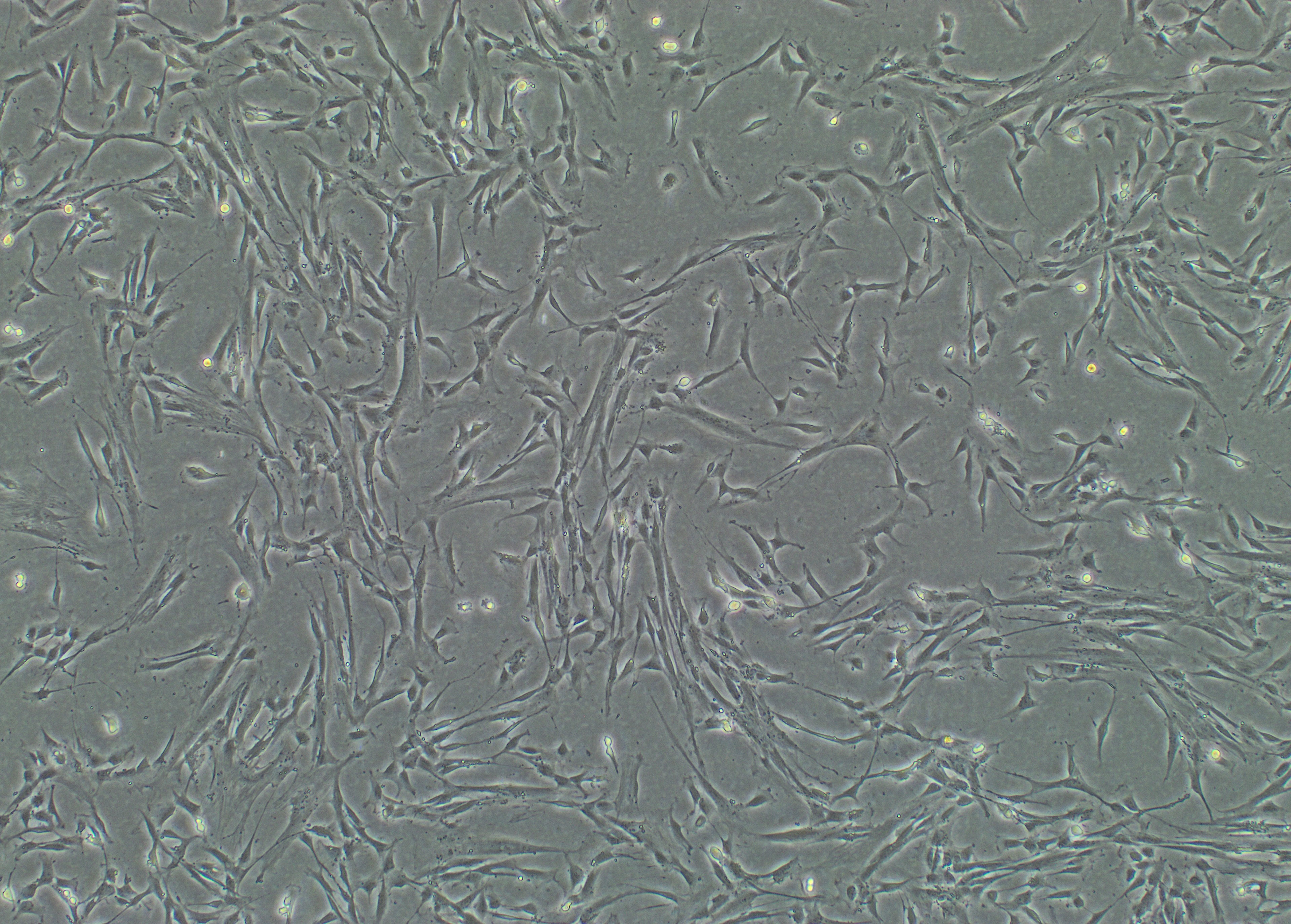
Foto: Human islets Foto: DSC ( Human decidual stromal cells )
