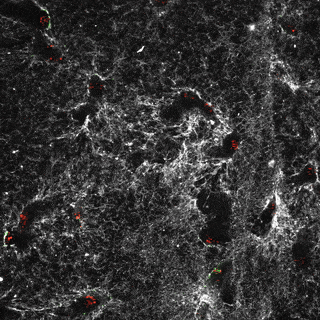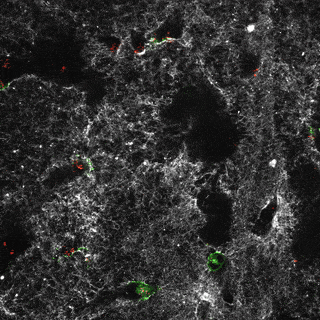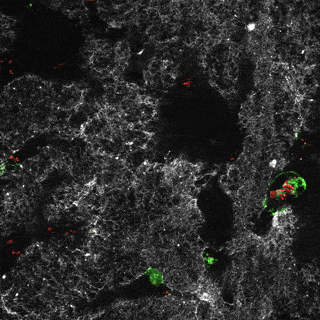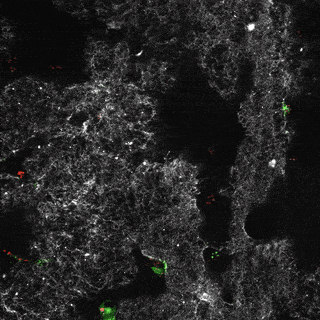
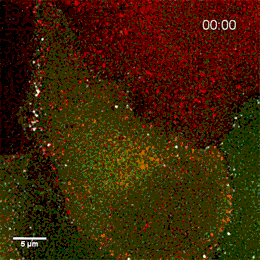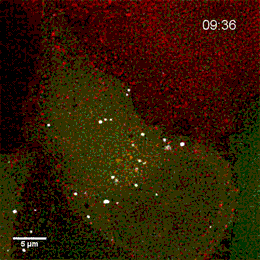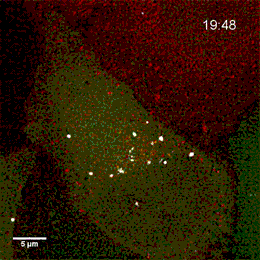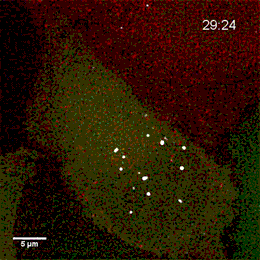
Project group leader Camilla Raiborg Protein and organelle dynamics in tumorigenesis

The transformation of a healthy cell into a cancer cell requires multiple events. Knowing the players involved in the processes that regulate cell growth, survival, differentiation, cell division and cell death, can in turn allow us to establish new strategies for diagnosis and treatment of cancers. In our research we focus on protein complexes that shape cellular membranes or alter cellular signalling and in this way contribute to tumour suppression or cancer development. Our aim is to uncover novel cellular mechanisms that can be exploited in cancer medicine.
|
|
|
Coworkers:
Funding:
 |
|
|